Stirling engine mug top
Concepts
I was reading about Stirling engines and they turn out to be pretty interesting. They have a number of properties that set them apart from common engines:
They rely on the difference in temperature between two plates
They operate on a closed loop: no fuel needed, no emission produced
That said you might need to fuel a secondary loop to generate your difference in temperature by heating a plate, but it means anything can be used there
They are very quiet since there is not any explosion or gas emission
They can be very efficient (up to 50% so comparable to diesel engines)
They are slow to start but steady once launched
Since they use temperature difference they are less efficient in hot places
Smaller engines tend to be more efficient than bigger ones
They are very same since there is no pressurized gas
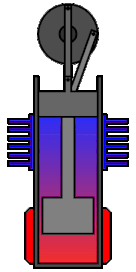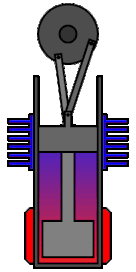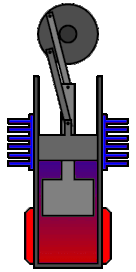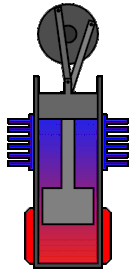
Clearly these are not fit for everything, but they have already found some applications especially when it comes to pumping wasted energy back in the system. For example some submarines use them by exploiting the temperature difference between the hot inside and cool waters in order to get back some of the energy that would otherwise heat the ocean.
Application
I was thinking of ways to use this at home and since I have a nice cup of tea before me it sounds like a good starting point. Let us consider a "mug top" that doubles as phone charger.
Now a mug contains about 0.3L of water, so about 300g. The energy the mug gives back to the room by cooling is equal to the energy we invested to heat it in the first place.
The energy needed to heat 1g of water by 1°C at 25°C is 1 calorie (by definition) or about 4 Joules. So heating our cup of tea from the ambient 25°C up to 95°C takes about
Therefore our hot cup of tea is a "battery" of 5000J. As we said, in perfect conditions our Stirling engine should be able to convert 50% of that energy to motion, but our tea cup does not present perfect conditions. Much heat escapes from the sides and our engine will not be a perfect one and we still need to convert that motion to electric current which adds another conversion layer.
Let us settle for about 30% efficiency in first approximation to convert heat to electricity. That represents
A standard iPhone 8 battery has a capacity of about 1800mAh according to internet. This will be a good starting point. We need to establish the link between our raw energy and battery capacity though.
We know that power = voltage × current, so energy / time = voltage × current and energy / voltage = current × time or in units J/V = A×s.
So to make the link we need to decide on a voltage. Standard 5V should do the trick. Then we have
These 94mAh represent 5% of our phone battery.

Conclusion
All of this means that, assuming we do not drink our tea until it cools back to room temperature, we can charge our phone by about 5%. This is not that great, but it is only using wasted energy which makes it essentially free. And maybe it would be more useful directly on the teapot or kettle. Damn, think of simply putting it on top of the lid every time you make pasta. Either way I do not think this energy is negligible when 4 teas a day represent 20% of a phone charge.
All in all I would like to see a future in which every household is filled with little power generators, either limiting energy waste or using green energies. This would probably not make centralized power plants obsolete but it could cut our expenses by quite a bit, especially when it comes to power dissipated when transporting the electricity from distant power plants to our homes.
Maybe it is worth taking the time to hack a 50€ Stirling engine together with an electric generator and see what comes out of this.

